
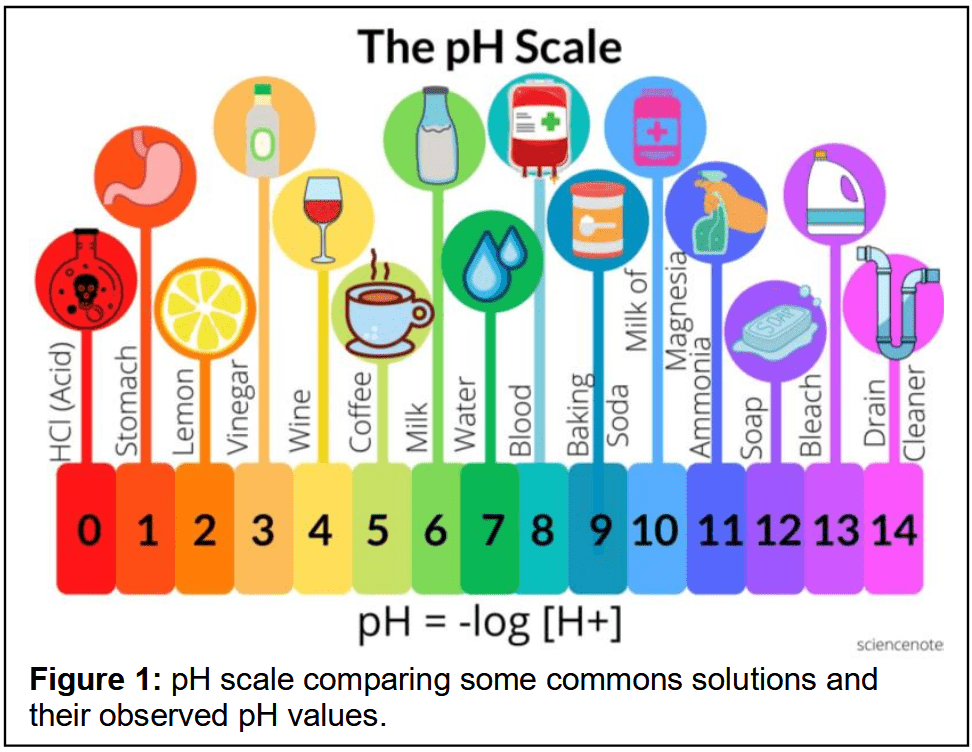
Most biological experiments require preparation of chemical solutions in their procedure. This is to ensure biomaterials are maintained at physiological conditions for measurements, as well as to alter the charge properties of biomolecules to allow efficient isolation using methods such as ion exchange chromatography or centrifugation. There are two principal methods for the preparation of these chemical solutions; by weight volume (w/v) and by volume volume (v/v). It is important to recognize the difference between these methods and the manner by which such solutions are prepared. Many experiments involving chemicals call for their use in solution form at a defined pH, Figure 1. That is, two or more substances are mixed together in known quantities and the pH is adjusted to a desired value. This may involve weighing a precise amount of dry material or measuring a precise amount of liquid and adjusting the pH accordingly. Preparing solutions accurately improves an experiment's safety and reproducibility.
Question
Why is it important to prepare chemical solutions accurately in biological experiments?
Answer the question above the continue reading. iTELL evaluation is based on AI and may not always be accurate.
Question
What is a buffer solution?
Answer the question above the continue reading. iTELL evaluation is based on AI and may not always be accurate.
The purpose of this procedure is to understand the basic principles of weighing solid, measuring liquids and adjusting the pH of solutions and to be able to conduct the process in the lab.
Question
What is the purpose of the procedure described in the passage?
Answer the question above the continue reading. iTELL evaluation is based on AI and may not always be accurate.
This procedure applies to qualified skills center users.
1.5 M Tris-HCl (pH 8.8)
Tris base 181.7g in 1L H2O, adjust pH to 8.8 using HCl.
0.5 M Tris-HCl (pH 6.8)
Tris base 60.6g in 1L H2O, adjust pH to 6.8 using HCl.
10% (w/v) SDS
SDS (sodium dodecyl sulfate) 100g in 1L H2O.
2X SDS Protein Sample Buffer
1.25M Tris HCl (pH 6.8), 4% (w/v) SDS, 20% (v/v) glycerol, trace bromophenol blue. Note: For reducing conditions use 950 ul 2x SDS sample buffer plus 50μL 2-mercaptoethanol.
10% (w/v) Ammonium Persulfate (APS)
10g ammonium sulfate in 100ml H2O, aliquot into 1ml vials and stored at –20ºC until needed.
Acrylamide/ bisacrylamide stock solution
For 1L, 30% (w/v) acrylamide 300g, 0.8 % (w/v) bisacrylamide 8g. Add H2O to the final volume and filter through the 0.22 μm membrane. Store at 4ºC in the dark.
Coomassie Brilliant Blue Staining Solution (1 liter)
Coomassie Brilliant Blue R250 2.5g, Methanol 400ml, Glacial Acetic Acid 100ml, H2O 500 ml
High Methanol Destain Solution
For 1L, Methanol 400ml, Glacial Acetic Acid 100ml, H2O 500ml
4% (v/v) Glycerol Solution
For 1L, Glycerol 40ml, H2O 960ml
Electrophoresis Buffer (TAE)
50X stock solution pH ~8.5, 242g Tris base, 57.1ml glacial acetic acid, 37.2g Na2EDTA.2H20, Add distilled water to 1 liter.
Solution 1. Using percentage by weight (w/v)
Formula, the formula for weight percent (w/v) is: [Mass of solute (g) / Volume of solution (ml)] x 100.
Example. A 10% NaCl solution has 10g of sodium chloride dissolved in 100ml of solution.
Procedure Weigh 10g of sodium chloride. Pour it into a graduated cylinder or volumetric flask containing about 80ml of water. Once the sodium chloride has dissolved completely (swirl the flask gently if necessary), add water to bring the volume up to the final 100ml. Caution: Do not simply measure 100ml of water and add 10g of sodium chloride. This will introduce error because adding the solid will change the final volume of the solution and throw off the final percentage.
Solution 2: Using percentage by volume (v/v). When the solute is a liquid, it is sometimes convenient to express the solution concentration as a volume percent.
Formula. The formula for volume percent (v/v) is: [Volume of solute (ml)/ Volume of solution (ml)] x 100.
Example. Make 1000ml of a 5% by volume solution of ethylene glycol in water.
Procedure. First, express the percent of solute as a decimal: 5% = 0.05. Multiply this decimal by the total volume: 0.05 x 1000ml = 50ml (ethylene glycol needed). Subtract the volume of solute (ethylene glycol) from the total solution volume: 1000ml (total solution volume) - 50ml (ethylene glycol volume) = 950ml (water needed). Dissolve 50ml ethylene glycol in a little less than 950ml of water. Now bring the final volume of solution up to 1000ml with the addition of more water. (This eliminates any error because the final volume of the solution may not equal the calculated sum of the individual components). So, 50ml ethylene glycol/ 1000ml solution x100 = 5% (v/v) ethylene glycol solution.
Solution 3: Molar Solutions. Molar solutions are the most useful in chemical reaction calculations because they directly relate the moles of solute to the volume of solution.
Formula. The formula for molarity (M) is: moles of solute / 1 liter of solution or gram-molecular masses of solute / 1 liter of solution.
The molecular weight of a sodium chloride molecule (NaCl) is 58.44, so one grammolecular mass
(=1 mole) is 58.44g. We know this by looking at the periodic table. The atomic mass (or weight) of Na is 22.99, the atomic mass of Cl is 35.45, so 22.99 + 35.45 = 58.44. Or more simply, just look at the container the chemical is sold in or on the Material Safety Data Sheet (MSDS) for molecular weight. If you dissolve 58.44g of NaCl in a final volume of 1 liter, you have made a 1M NaCl solution, a 1 molar solution.
Procedure. To make molar NaCl solutions of other concentrations dilute the mass of salt to 1000ml of solution as follows: 0.1M NaCl solution requires 0.1 x 58.44 g of NaCl = 5.844g. 0.5M NaCl solution requires 0.5 x 58.44 g of NaCl = 29.22g. 2M NaCl solution requires 2.0 x 58.44 g of NaCl = 116.88g
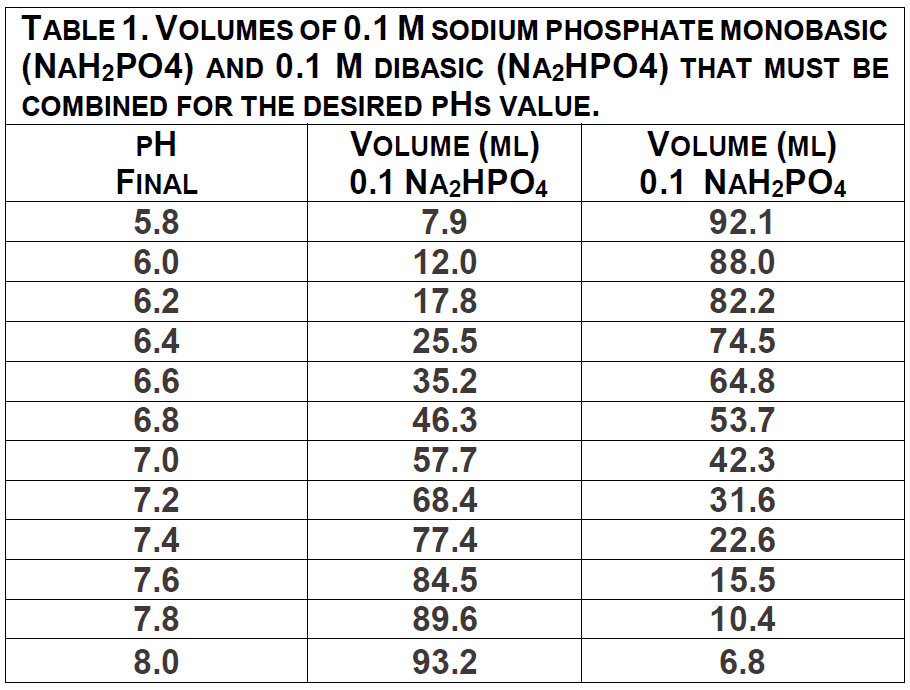
Solution 4: Sodium Phosphate buffer. Some buffers are prepared by adding two equal concentration solutions of similar types together to reach the proper buffering range and pH. The preparation of a sodium phosphate buffer is one example of this method.
Formula. 0.1M monobasic (NaH2PO4) and 0.1M dibasic (Na2HPO4) sodium phosphate solutions must be prepared first (See recipes above). To reach the desired pH of a 0.1M sodium phosphate buffer, follow recipe in Table 1. 7.15. Example. You need 100mL of a sodium phosphate buffer pH 6.7. Prepare 0.1M solutions of both mono- and di-basic sodium phosphate (See recipes). Add 565
ml 0.1 M NaH2PO4 to 435 ml 0.1 M Na2HPO4 in a sterile flask.
Procedure. Prepare 0.1M solutions of both mono- and di-basic sodium phosphate as directed in recipes. Determine desired pH of final buffer. Add appropriate amounts of mono- and di- basic sodium phosphate to final volume of 1L in sterile flask according to the lookup table.
Question
How can you address the issue of not being able to weigh out very small quantities of buffer components?
Answer the question above the continue reading. iTELL evaluation is based on AI and may not always be accurate.
This task will test your ability to make several standard laboratory buffers with the
correct pH.
Create a table showing the quantities (volume/ weight) of every component of the
following buffers:
Buffer 1. 50 mL of 0.2M K2HPO4, 100mM NaCl, pH 7.9
Buffer 2. 25 mL of 0.1M TRIS, 50 mM NaCl, pH 8.1
Buffer 3. 40 mL of 0.1M HEPES, 80mM NaCl, 1% glycerol, pH 7.4
Buffer 4. 50 ml of 0.1 M Sodium Phosphate buffer, pH 7.0 made from stock 0.1M
NaH2PO4 and 0.1M Na2HPO4 solutions using Table 1.
Make each of the buffers listed above and store in a 50 mL conical tube, label them with your name, date and the buffer components. Paste a table Provide a listing the components and the various amounts in each buffer. Once you submit your MMT with your table of calculated components a proctor will test your buffers.
Last updated at